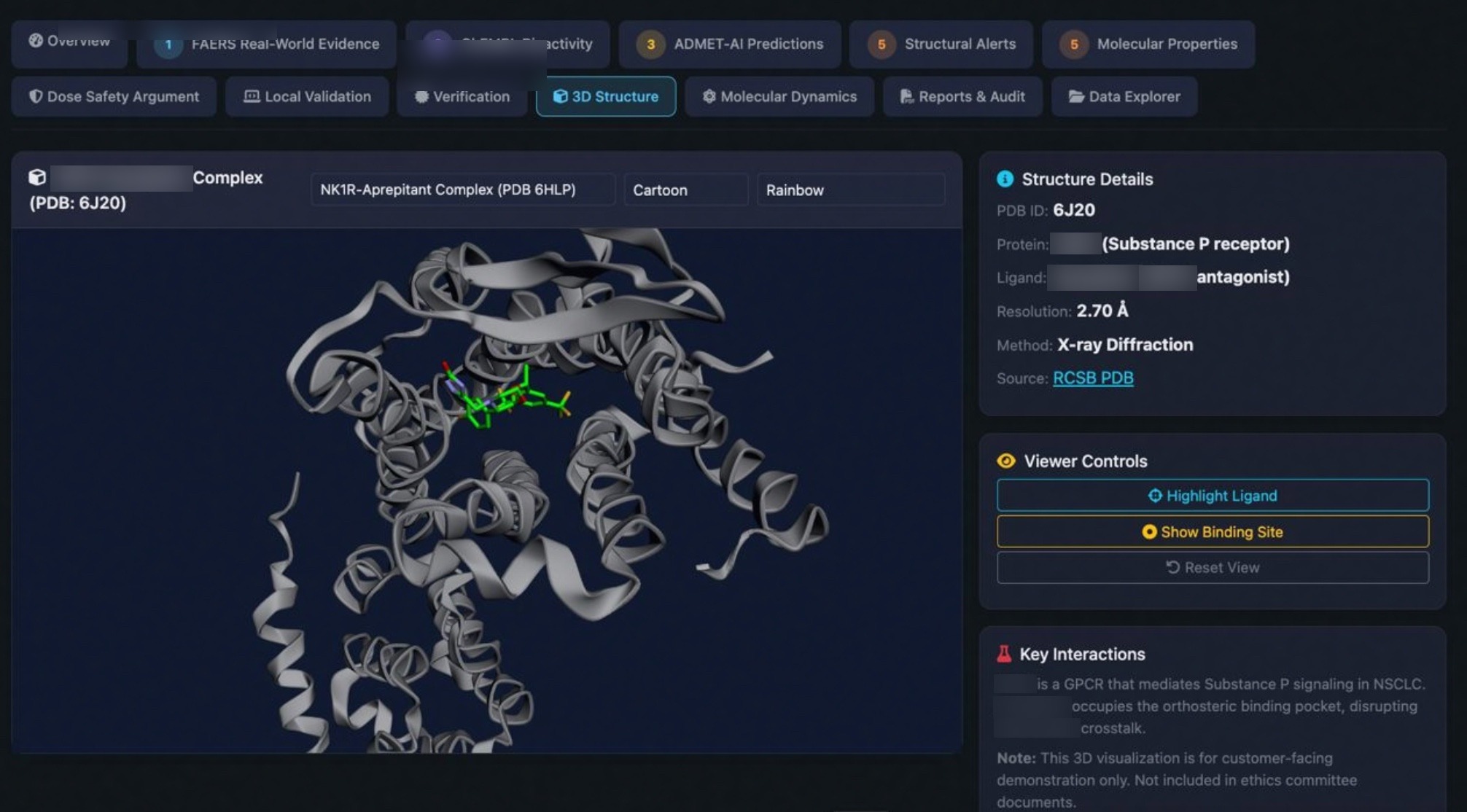
Research Credibility
20+ Bio Papers at World-Class Venues
Our Alzheimer's and bio research validated at the same conferences where Google DeepMind, Genentech, and Stanford publish. Our founder has been an AI researcher since the 1990s.
AAAI-26
5
Drug discovery & Alzheimer's AI — AIDD & LMReasoning workshops. 1 oral presentation.
#4 Google Scholar · h5-index 212
Top AI conference since 1979. Same stage as OpenAI, DeepMind & Stanford AI Lab. 17,000+ researchers attend.
Official AAAI site →
ICLR 2026
5
Biomedical AI, drug discovery & world models workshops.
#2 Google Scholar · h5-index 304
World's premier deep learning conference. Fully open peer-review. Where Google Brain, Meta FAIR & Genentech AI publish.
Official ICLR site →
NeurIPS
1
AI for Drug Discovery Workshop — computational biology focus.
#1 Google Scholar · h5-index 337
The world's most-cited AI conference. 15,000+ submissions, ~25% acceptance. Every major AI lab on Earth publishes here.
AI4D3 Drug Discovery Workshop →
MLGenX
3
Generative AI for molecular drug discovery — Alzheimer's targets.
ICLR-affiliated · inherits #2 world ranking
Workshop at ICLR 2026 focused on generative AI for experimental sciences and molecular discovery.
MLGenX workshop →
AI4X-AC
6
Applied AI for science — 2 oral presentations on Alzheimer's & cancer.
Peer-reviewed AI × Science
International AI for Science conference held in Ljubljana, Slovenia. Bridges AI with biology, chemistry & medicine. Rigorous peer review.
Conference site →
ELLIS
1
ML4Molecules Workshop — molecular drug discovery.
Europe's top AI research network
European Laboratory for Learning & Intelligent Systems — Europe's equivalent to MIT's AI Lab. Members include Yoshua Bengio (Turing Award).
ML4Molecules workshop →
Stanford
1
Agents4Science Workshop — AI agents for scientific discovery.
Stanford University · Nobel laureate advisory
Hosted by Stanford University, one of the world's top research institutions. Workshop on autonomous AI scientists — the frontier of AI-driven discovery.
Stanford site →